Amyloid diseases, including Alzheimer’s, Parkinson’s, type-2 diabetes and other life-threatening diseases, involve pathologic deposits of normally soluble proteins or peptides as insoluble amyloid fibrils. When this happens in vital organs, such as the brain, kidney, liver and heart, it causes organ damage and, if left untreated, death. Unfortunately, the available treatment options are very limited.
Now a new study from researchers at Boston University School of Medicine (BUSM) improves our understanding of how heparan sulfate and related biopolymers such as heparin, which is perhaps best known as a blood thinner, can promote amyloid deposition in various organs.
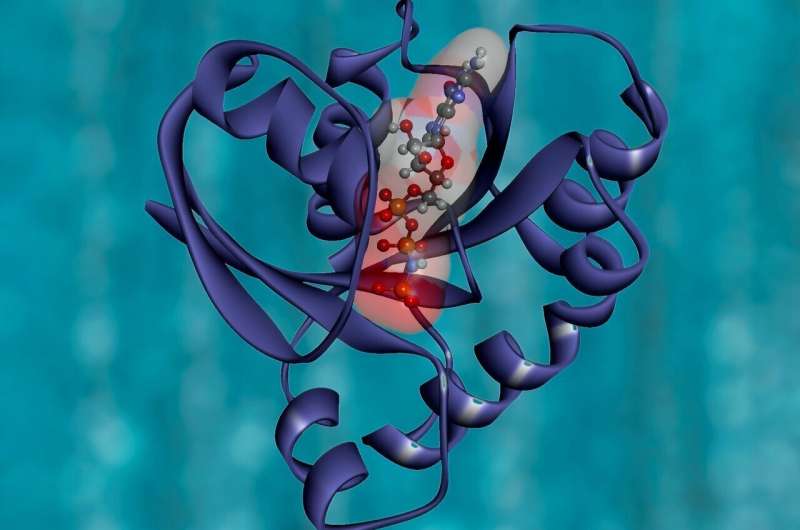
The researchers explain how charged biopolymers that cover the cell surfaces, such as heparan sulfate proteoglycans, trigger the formation of pathologic amyloids in deadly human diseases including Alzheimer’s disease, chronic traumatic encephalopathy, inflammation-linked amyloidosis and many others. “Heparan sulfate has long been known to accelerate amyloid aggregation in diverse proteins. Why this happens was unclear. We proposed the first detailed structural model that shows how heparan sulfate can aid protein aggregation in amyloid,” explained corresponding author Olga Gursky, Ph.D., professor of physiology and biophysics at BUSM.
According to the researchers, molecules such as heparan sulfate are not innocent bystanders but, rather, active participants in amyloid deposition in the body. “Interactions of these molecules with amyloid have been proposed as therapeutic targets. By understanding how charged biopolymers interact with amyloid aggregates on a molecular level, we can begin to design inhibitors to block these pathologic interactions,” said Emily Lewkowicz, Ph.D. student in biophysics and the lead author of this study.
The researchers used spectroscopy and electron microscopy to explore protein-heparin interactions. This included proteins such as oxidized human apoA-I, which forms amyloid in cardiovascular disease. Their analysis showed that heparin speeds up amyloid nucleation and growth by this and other proteins. To explain why this happens, they then performed computational studies using atomic structures of amyloid fibrils and found that patient-derived amyloids contain bound heparin-like molecules associated with deadly human diseases such as Alzheimer’s or chronic traumatic encephalopathy.
Amyloid diseases are incurable disorders that afflict hundreds of millions worldwide and represent a major public health challenge of the 21st century. Research such as this will help guide targeted drug design for these deadly diseases. “Therapeutic targeting of amyloidoses requires a clear understanding of factors that promote or prevent amyloid deposition. This research brings us one step closer to designing amyloid-specific therapies,” added Lewkowicz.
Source: Read Full Article