Vaccinations have been rolling out in many parts of the world since December 2020 in an attempt to turn the tide of the coronavirus disease (COVID-19) pandemic. But are COVID-19 vaccines safe for pregnant women?
New research by a team at Northwestern University Feinberg School of Medicine, USA, suggests that the COVID-19 vaccine does not damage the placenta and may be safe to use during pregnancy.
The study, published in the American Journal of Obstetrics and Gynecology, is the first study to assess the effect of mRNA COVID-19 vaccines on the placenta.
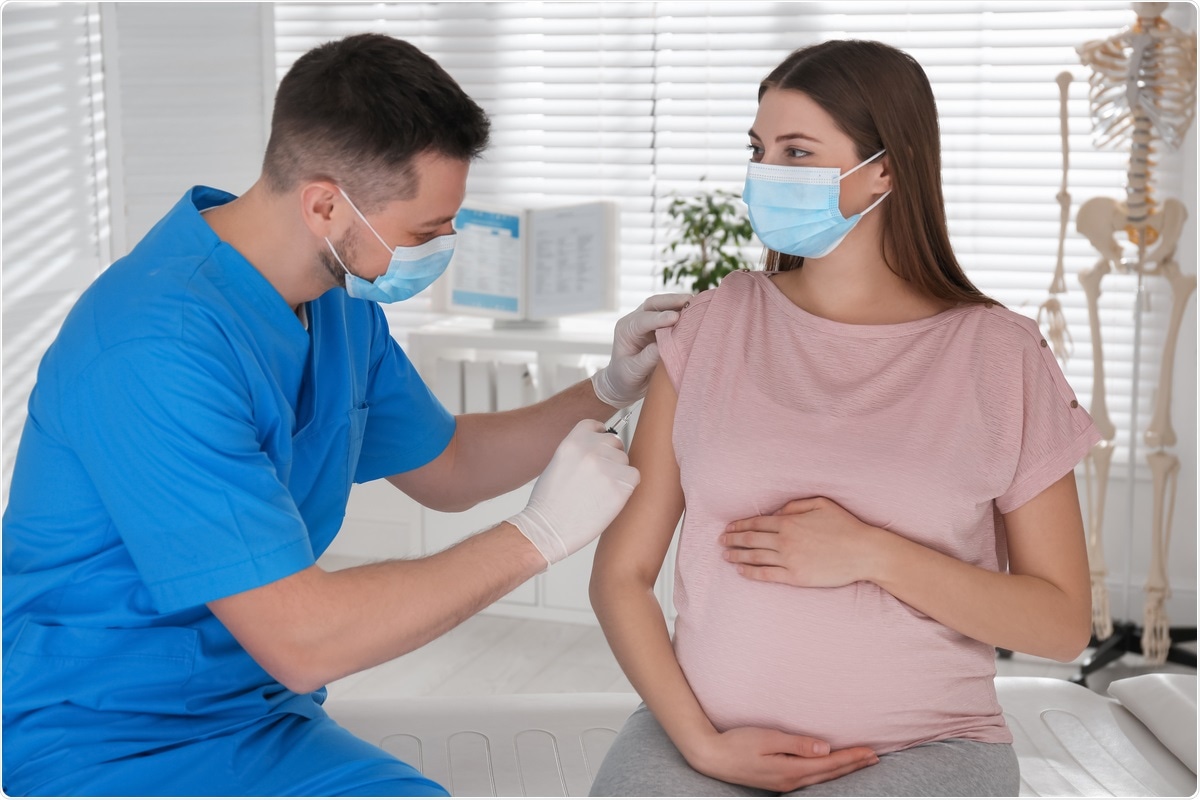
Placenta and vaccines
The placenta is a crucial element in pregnancy. It is akin to an aircraft’s “black box,” explains Dr. Jeffery Goldstein, a co-author in the study. If something goes wrong during pregnancy, placental changes can help determine the problem.
The World Health Organization (WHO) reports that vaccine-preventable contagious infections are responsible for significant maternal, neonatal, and young infant morbidity and mortality.
The changes in the immune response of pregnant women, which happens to prevent the immune system from attacking the fetus, may interfere with the development of immune responses to pathogens.
Hence, pregnant women are at a higher risk of infections due to a suppressed immune system.
For instance, when the influenza vaccine is given to pregnant women, it may also protect the unborn child. This means that the vaccine is safe and well-tolerated for pregnant women.
However, among the various vaccines developed, the attenuated rubella and mumps viruses can enter the placenta and cause complications. The measles, mumps, and rubella vaccine (MMR) is therefore not recommended to be given to pregnant women.
The various COVID-19 vaccines have yet to be examined for adverse effects, especially as these are newly developed. A previous study, however, found that messenger RNA (mRNA) vaccines are generally safe to use in pregnant women. The Moderna and BioNTech-Pfizer COVID-19 vaccines are both based on an mRNA platform.
COVID-19 vaccine during pregnancy
The current study aimed to investigate the transfer of SARS-CoV-2 immunoglobulin G (IgG) antibodies to infants following maternal COVID-19 vaccination during pregnancy, and the factors tied to increased efficiency of the transfer.
The researchers identified pregnant women who delivered at the Prentice Women’s Hospital in Chicago, Illinois, from January 2021 to March 2021. They also identified those who received the COVID-19 vaccine. Overall, the study involved 84 vaccinated pregnant women and 116 unvaccinated patients.
At the time of the study, two mRNA COVID-19 vaccines – the Pfizer-BioNTech and Moderna vaccines – had been granted emergency use authorization in the USA.
From there, the team collected maternal and umbilical cord blood from samples submitted for blood typing.
The researchers measured the SARS-CoV-2 IgM and IgG antibody levels from participants’ plasma along with antibody transfer ratios.
The researchers noted that placentas of women who tested positive for COVID-19 while pregnant showed placental injury, marked by the abnormal flow between the mother and fetus in utero. Meanwhile, the researchers observed no injury to the placenta of vaccinated women who gave birth.
The team also looked for abnormal blood flow between the mother and fetus and issues with fetal blood flow. The study did not find any cases of chronic histiocytic intervillositis, a complication that can happen if the placenta is infected, in vaccinated patients.
Further, most pregnant women who received the COVID-19 vaccine during the third trimester had a transplacental transfer of IgG to the infant. This means that antibodies were transferred from the mother to the unborn child, providing dual protection.
The observed IgG transfer ratio showed that the antibody levels in the infant were about equal to maternal levels.
These data suggest, at least among women in their third trimester, that earlier vaccination may produce a greater infant immunity, the immunobiology of which requires further study,” the team concluded.
They added that future research is needed to validate the results and should include a more diverse cohort of women.
Nevertheless, these results show promising evidence for passive immunity against SARS-CoV-2 in newborns after maternal receipt of COVID-19 mRNA vaccinations,” they added.
- Supriya, Lakshmi. (2021, April 23). Study reports on safety of COVID-19 mRNA vaccines in pregnancy. News-Medical. Retrieved on May 13, 2021 from https://www.news-medical.net/news/20210423/Study-reports-on-safety-of-COVID-19-mRNA-vaccines-in-pregnancy.aspx
- World Health Organization (WHO). (2014). Safety of immunization during pregnancy. https://www.who.int/vaccine_safety/publications/safety_pregnancy_nov2014.pdf
- Mithal, L., Otero, S., Shanes, E. et al. (2021). Cord blood antibodies following maternal coronavirus disease 2019 vaccination during pregnancy. The American Journal of Obstetrics and Gynecology. https://doi.org/10.1016/j.ajog.2021.03.035, https://www.ajog.org/article/S0002-9378(21)00215-5/fulltext
Posted in: Child Health News | Medical Science News | Medical Research News | Women's Health News | Disease/Infection News | Healthcare News
Tags: Antibodies, Antibody, Blood, Chronic, Coronavirus, Coronavirus Disease COVID-19, Gynecology, Hospital, Immune Response, Immune System, Immunoglobulin, In Utero, Influenza, Measles, Medicine, Messenger RNA (mRNA), MMR, Mortality, Mumps, Obstetrics, Pandemic, Placenta, Pregnancy, Research, RNA, Rubella, SARS, SARS-CoV-2, Umbilical Cord, Vaccine, Virus

Written by
Angela Betsaida B. Laguipo
Angela is a nurse by profession and a writer by heart. She graduated with honors (Cum Laude) for her Bachelor of Nursing degree at the University of Baguio, Philippines. She is currently completing her Master's Degree where she specialized in Maternal and Child Nursing and worked as a clinical instructor and educator in the School of Nursing at the University of Baguio.
Source: Read Full Article